API & Intermediates
Your Trusted Partner in API and Intermediates Manufacturing
Maithili Life Sciences is a renowned name in the pharmaceutical industry, revered for its commitment to excellence in the manufacturing of Active Pharmaceutical Ingredients (APIs) and intermediates. As a leading API manufacturer in India, we pride ourselves on our ability to deliver high-quality, innovative products that cater to the evolving needs of the global healthcare market.
Our Commitment to Quality
At Maithili Life Sciences, quality is our cornerstone. Our meticulously manufactured APIs and Intermediates adhere to the highest international standards, ensuring the efficacy, safety, and reliability of the final pharmaceutical formulations. With a relentless commitment to quality control and regulatory compliance, we stand as your trusted partner in delivering pharmaceutical solutions that make a difference.
Comprehensive Portfolio of API and Intermediates
Dive into our comprehensive portfolio, meticulously crafted to meet the diverse needs of the pharmaceutical industry:
API Excellence
Our validated APIs, including Dabigatran Etexilate Mesylate, Pregabalin, Sumatriptan, and more, represent the pinnacle of pharmaceutical innovation. Rigorous testing and adherence to global pharmacopeias guarantee the highest quality standards.
Intermediates Expertise
Maithili Life Sciences excels in the production of intermediates critical to pharmaceutical synthesis. Our commitment to precision and consistency ensures seamless integration into your manufacturing processes.
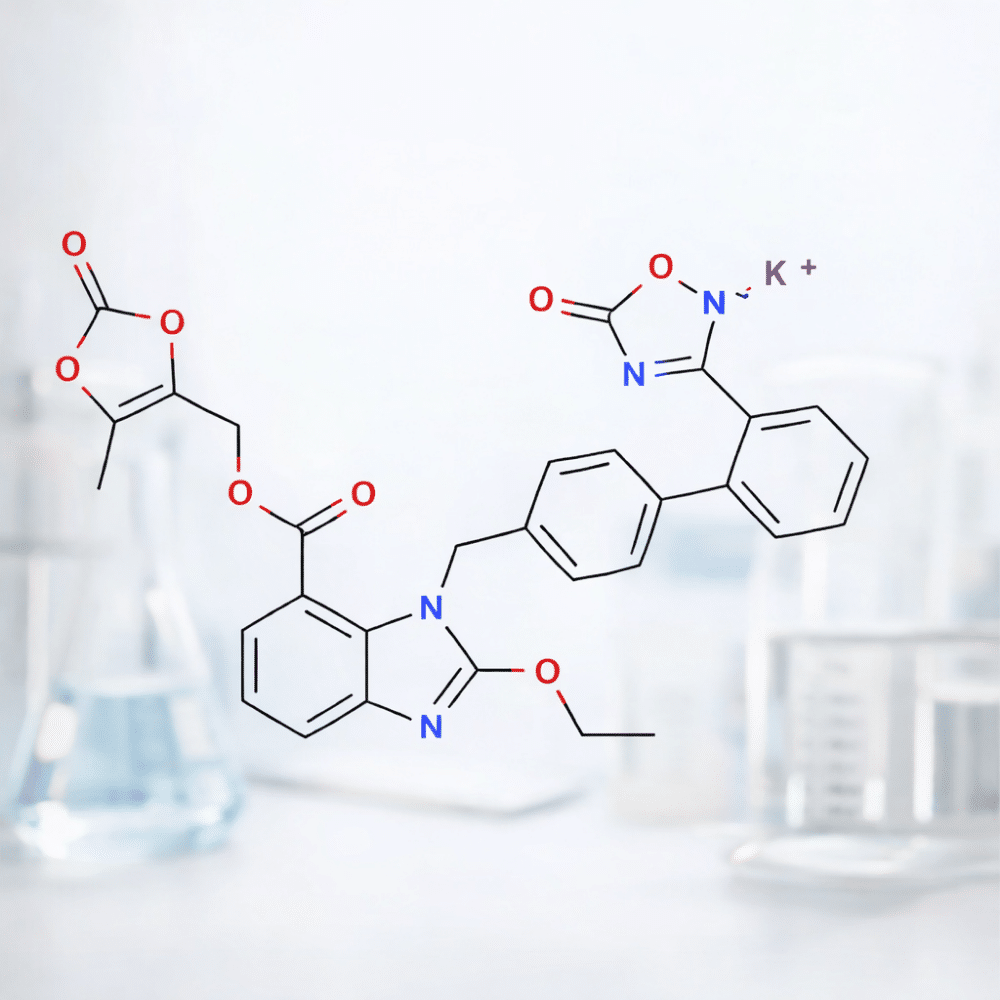
Azilsartan Medoxomil Potassium Salt
Apply Now
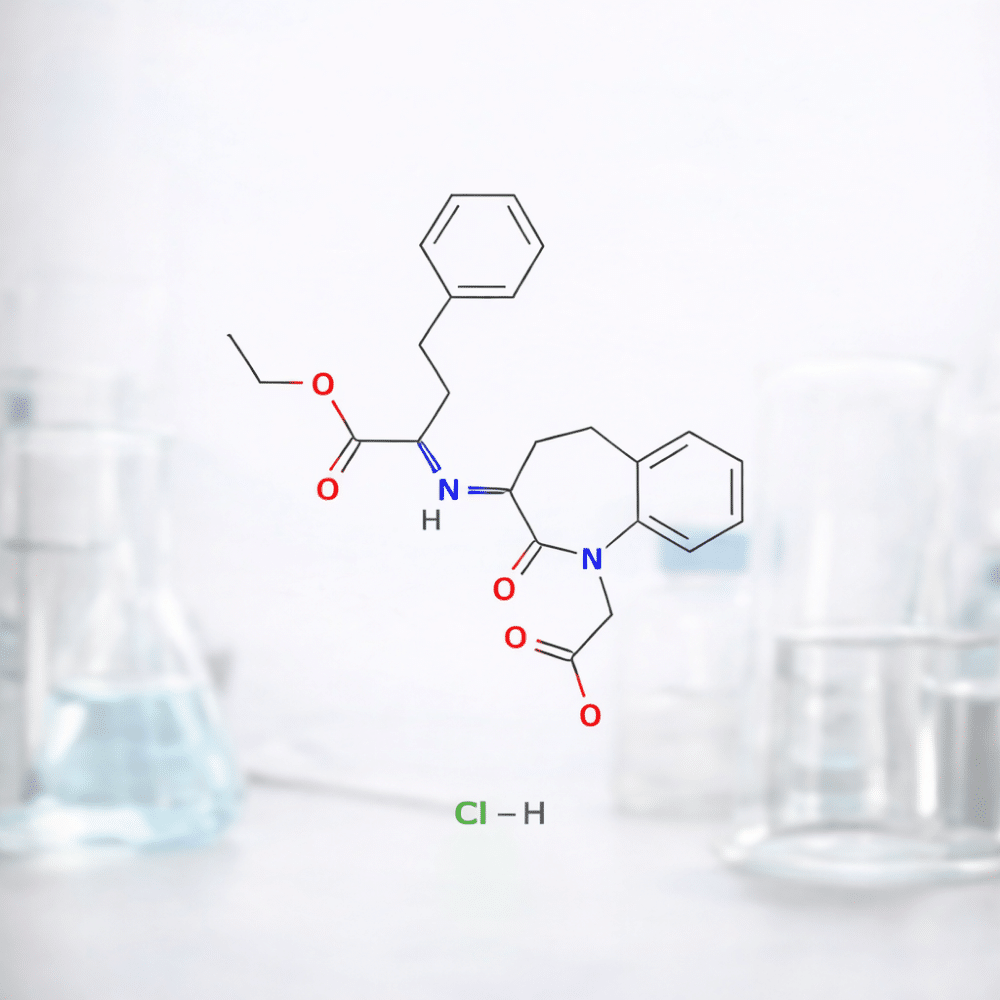
Benazepril Hcl
Apply Now
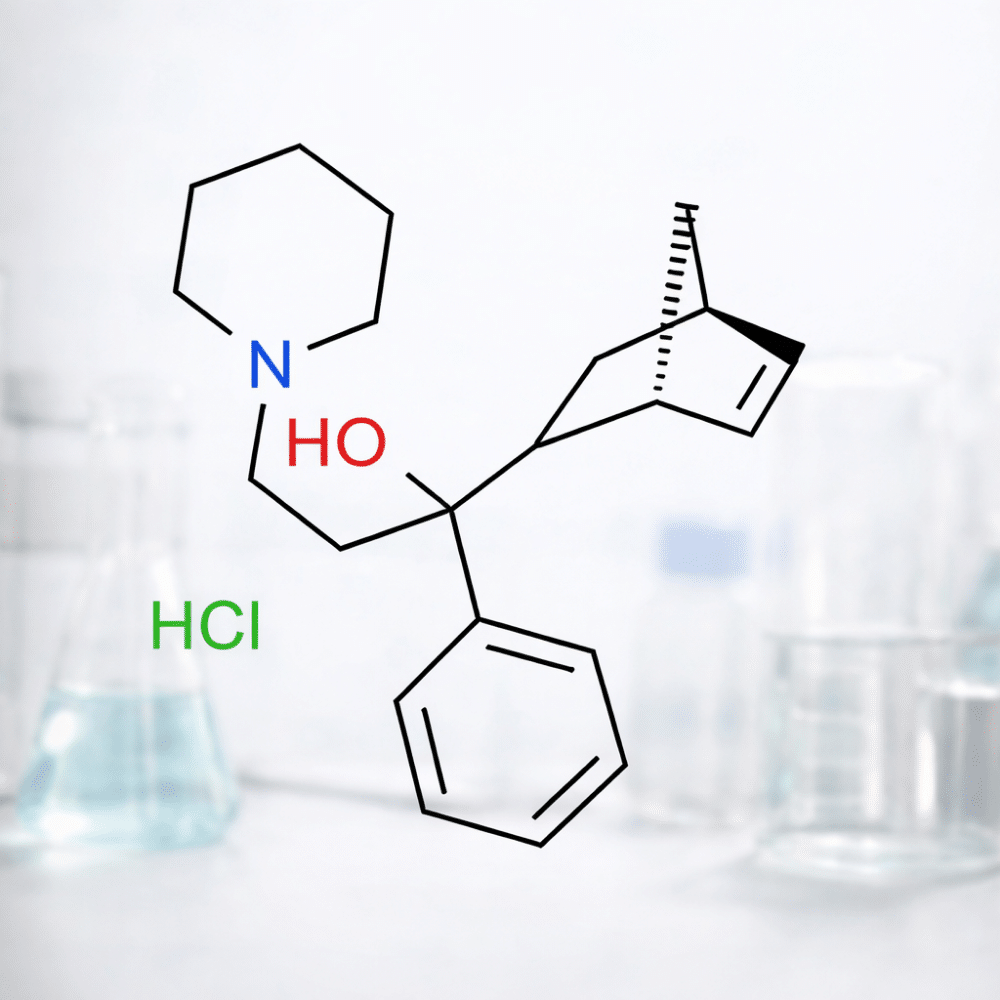
Biperiden Hcl
Apply Now
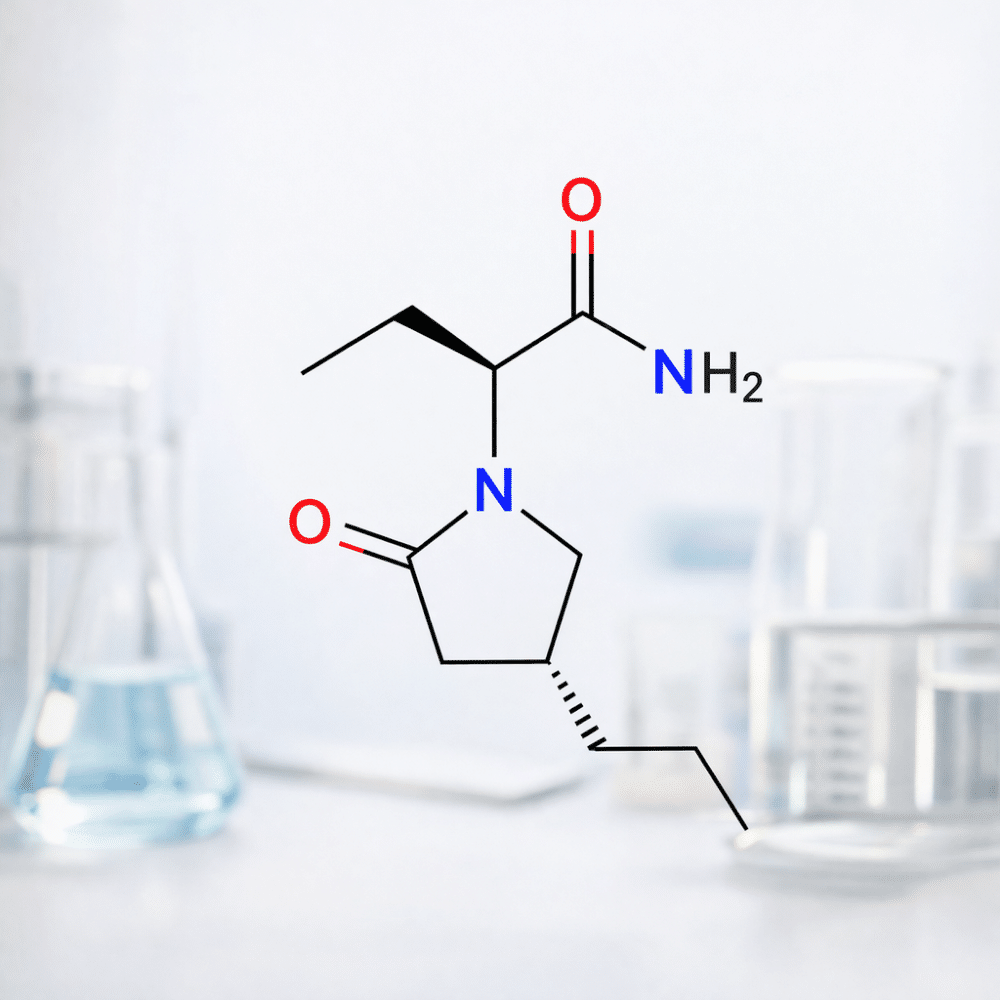
Briveracetam (R&D)
Apply Now
Cabergoline
Apply Now
Darolutamide (R&D)
Apply Now
Dabigatran Etexilate Mesylate
Apply Now
Dapoxetine Hcl
Apply Now
Enclomiphene Citrate (R&D)
Apply Now
Favipiravir
Apply Now
Irbesartan
Apply Now
Minoxidil
Apply Now
Modafinil
Apply Now
Naftidrofuryl Oxalate
Apply Now
Nebivolol HCl
Apply Now
Ornidazole
Apply Now
Rasagiline Mesylate
Apply Now
Solifenacin Succinate
Apply Now
Sumatriptan Succinate
Apply Now
Tadalafil
Apply Now
Tamsulosin
Apply Now
Telmisartan
Apply Now
Torsemide
Apply Now
Trazodone Hcl
Apply Now
Ticagrelor
Apply Now
Vardenafil Hcl Trihydrate
Apply Now
Voriconazole
Apply Now
Zaltoprofen
Apply Now
Innovation as a Driving Force
At Maithili Life Sciences, we don’t just follow industry trends; we set them. Our state-of-the-art research and development facilities are hubs of innovation, constantly pushing the boundaries of pharmaceutical science. By investing in cutting-edge technologies, we deliver not just products but solutions that anticipate and meet the evolving demands of the industry.
Sustainability at the Core of Our Practices
We recognize the responsibility that comes with our role in healthcare. Sustainability is not just a buzzword for us; it’s a commitment embedded in our practices. From eco-friendly manufacturing processes to ethical sourcing, we prioritize environmental responsibility. Partnering with Maithili Life Sciences means aligning with a company that cares not only about your success but also the well-being of our planet.
Why Choose Maithili Life Sciences for your API and Intermediates?
Global Reputation
With a strong presence in the international pharmaceutical landscape, Maithili Life Sciences is your global partner in healthcare advancement.
Quality Assurance
Our APIs and Intermediates undergo stringent quality control measures, ensuring a consistent and reliable supply for your manufacturing needs.
Collaborative Approach
We believe in collaborative success. Your goals become ours, and together, we drive innovation and excellence in pharmaceutical manufacturing.
Contact Us for a Transformative Partnership
Embark on a transformative journey with Maithili Life Sciences. Whether you seek high-quality APIs, intermediates, or a strategic collaboration, we invite you to explore the possibilities with us. Reach out to our team today, and let’s shape the future of healthcare together.
Kindly Note
Products protected by valid patents are not offered for sale in countries where the sale of such products constitutes a patent infringement and its liability is at buyer’s risk.